Alzheimer’s Disease: A Phase 2b Liraglutide Trial Protocol & Population
Liraglutide Shows Promise in Slowing Alzheimer’s Progression, Phase 2 Trial Suggests
LONDON – A new study published today offers a glimmer of hope in the fight against Alzheimer’s disease, a condition that currently affects over 55 million people worldwide, according to the World Health Organization. Researchers have found that liraglutide, a medication already approved for type 2 diabetes and obesity, may slow the progression of cognitive decline in individuals with early-stage Alzheimer’s. The findings, from a Phase 2b clinical trial conducted across the United Kingdom, suggest a potential new avenue for treatment, though experts caution that further research is needed.
A Novel Approach to Alzheimer’s Treatment
For decades, Alzheimer’s research has largely focused on targeting amyloid plaques and tau tangles – the hallmark proteins that accumulate in the brains of those with the disease. However, recent failures of drugs aimed directly at these targets have prompted scientists to explore alternative strategies. This trial investigated liraglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, which has shown neuroprotective effects in animal models. The rationale behind using liraglutide stems from its ability to reduce inflammation and potentially improve brain metabolism, factors increasingly recognized as playing a role in Alzheimer’s development.
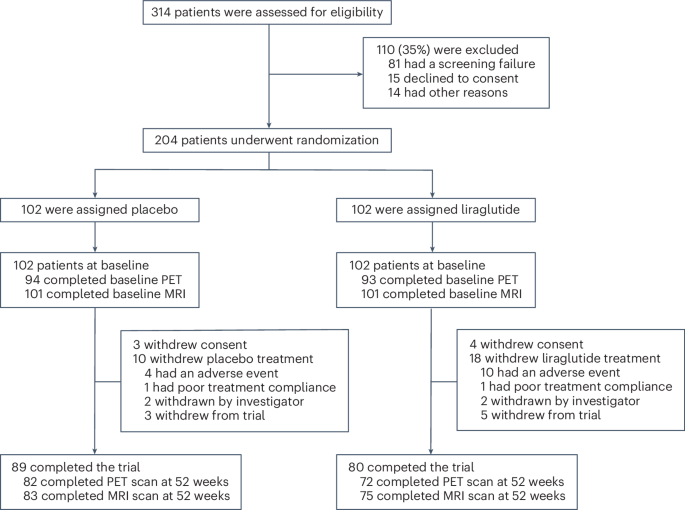
The 12-month, double-blind, placebo-controlled trial involved 204 participants aged 50 and older with mild to moderate Alzheimer’s disease. Participants were recruited from memory clinics and specialized research networks across the UK. They were randomly assigned to receive either liraglutide or a placebo, with doses escalating from 0.6mg to a maximum of 1.8mg daily, adjusted based on individual tolerance. The study meticulously tracked cognitive function, brain metabolism using PET scans, and brain volume changes via MRI.
Key Findings: Improved Brain Metabolism and Cognitive Stability
The primary outcome of the study focused on changes in cerebral glucose metabolism, a measure of brain activity, assessed using [18F] FDG PET scans. Researchers observed a statistically significant improvement in glucose metabolism in key brain regions – the hippocampus, medial temporal lobe, and posterior cingulate cortex – in the liraglutide group compared to the placebo group. This suggests that the drug may help restore energy production in brain cells affected by Alzheimer’s.
While cognitive scores didn’t show a dramatic improvement, the study found that participants receiving liraglutide experienced a slower rate of cognitive decline as measured by the ADAS-Exec, a composite cognitive assessment tool. This composite score combines elements of the traditional ADAS-Cog with tests focusing on executive function, addressing some of the limitations of earlier cognitive assessments. MRI scans also revealed a slower rate of ventricular enlargement – a sign of brain atrophy – in the liraglutide group.
Safety and Tolerability: A Favorable Profile
Liraglutide was generally well-tolerated by participants. The most common side effects were mild gastrointestinal issues, consistent with the known side effect profile of the drug when used for diabetes or obesity. The study adhered to rigorous safety protocols, including regular monitoring of vital signs, laboratory tests, and electrocardiograms. An independent data monitoring committee and steering committee oversaw the trial to ensure participant safety and data integrity.
Implications for Future Research and Treatment
Dr. Livia Grant, Senior Health Editor at worldys.news, commented on the significance of the findings: “This study is encouraging because it explores a completely different mechanism for potentially treating Alzheimer’s. The fact that a drug already approved for other conditions shows promise in this area is a significant advantage, potentially accelerating its path to clinical use if further trials confirm these results.”
The researchers emphasize that this is a Phase 2b trial, meaning it was designed to assess the drug’s safety and potential efficacy, not to definitively prove its effectiveness. Larger, Phase 3 trials are now needed to confirm these findings and determine the optimal dose and duration of treatment. These trials will also need to investigate whether liraglutide can delay the onset of dementia in individuals at high risk of developing the disease.
The study’s findings are particularly relevant given the growing global burden of Alzheimer’s disease. According to the Alzheimer’s Association, one in nine Americans aged 65 and older has Alzheimer’s disease, and this number is projected to rise dramatically as the population ages. The development of new and effective treatments is therefore a critical public health priority.
The open-label extension of the trial, where all participants received liraglutide, is ongoing, and its results are eagerly awaited. Researchers are also planning to analyze genetic data and plasma biomarkers collected during the trial to identify potential predictors of treatment response.
This research offers a renewed sense of optimism in the search for effective Alzheimer’s treatments, highlighting the importance of exploring novel therapeutic strategies and repurposing existing drugs.